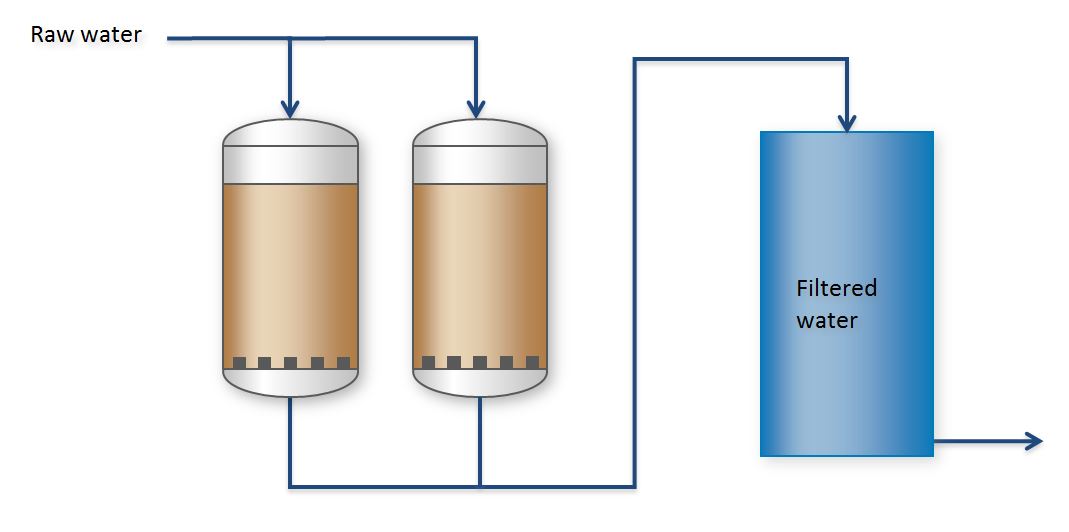
Classical water filtration

Classic alternatives to this modern membrane separa tion are gravel and multi-layer filters. Gravel or multi-layer filters are used to remove suspended solids, caused either by a previous oxida tion process, through defective pipes or by flocculation or precipitation.
Sandfilter
The most commonly employed filter uses gravel, in w hich various types and sizes of sand and gravel are laye red. If the iron content of the raw water is very high, a precipitation reactor is installed prior to the fil tration. The use of a flocculating agent, in most cases poly - aluminium chloride (PAC), will agglomerate the iron so that it can easily be removed by the filter.
The use of lime is also possible, since this increases the pH value and it also serves as a seed crystal for the flocculation process. In contrast, chlorine has an inhibitory effect on t he formation of flocs.

Interested?
At EUWA, good service is of high priority.
That's why we would like to advise you personally and competently. Make your request here and we will get back to you as soon as possible.
Multi-layer filters
Multilayer filters consist of several types of sand , gravel and hydro-anthracite. Hydro-anthracite is a carbona ceous material that allows an in-depth filtration. The pa rticles are filtered, not only at the surface but also in t he bed itself. Accordingly, the cleaning capacity is highe r and less frequent backwashing is required. These multi-layer filters also facilitate H2S removal. Both gravel and multilayer filters need to be back flushed with air. In the case of sand filters the backwash process takes place in combination with water, but for mult i-layer filters however it needs to take place separately.
If the water contains arsenic, it can be removed wi th GFH-filters (Granular ferric hydroxide) containing synthetic granular ferric hydroxide. It is an adsor bent having a high porosity and a large internal surface . The material is relatively expensive and must be replac ed when it is exhausted.
Therefore, it should not be used for particle filtr ation, since this reduces the adsorption capacity of GFH, which leads to higher costs. If elevated manganese levels are present in raw wat er, so called Greensand can be applied as filtration material whi ch catalytically supports the oxidation of manganese a nd its subsequent removal through filtration. It is a natu rally mined granular filtration material with a significant con tent of manganese oxide. This manganese oxide acts as a cat alyst for the oxidation of manganese dissolved in water. Prerequi site to complete this oxidation process is a saturation of oxygen in the water to be treated and a pH of at least 6,5.
In order not to disturb the catalytic oxidation of dissolved manganese on the Greensand surface, iron and turbid ity need to be removed first as much as possible if present in water. At higher iron and turbidity levels removal of iron an d turbidity maybe required in a separate filter upstream of the Greensand filter.
Illustration of classical filtration
Downloads
Here you will find current downloads with further information.